|
The higher the dew point, the higher will be the humid atmosphere. It is because no more water vapors are required. The dew point describes the saturation point of the vapors, where the air is at the saturation state and cannot hold more water vapors. Now, we must be confused about the relation between the dew point and the relative humidity of the air. RH is the relative humidity Dew point vs. RH is the relative humidity Dew point formula in degrees Fahrenheit The dew point formula when the actual air temperature and the relative humidity are given is: Here, we will discuss the dew point formula in terms of both degrees Celsius and degrees Fahrenheit. The above data clearly depicts that the higher the temperature, the higher will be the humidity percentage in the air. The table shows the humidity percentage at a different temperature range in Celsius as well as Fahrenheit. The humidifier is used to add moisture to the air when the air is dry, and the dehumidifier is used to remove the moisture from the air. The dew point of the air can also be controlled using a combination of sensors, a humidifier, and a dehumidifier. 
It is commonly known to measure water vapors or the moisture in the air and other places. The device used to measure the dew point temperature is a hygrometer. Humidity percentage at different temperatures The atmosphere at different dew points is listed in the below table: Dew point range The higher the dew point, the more moisture the air would contain, which means the air's humidity increases. We can also say that air is heavy and has no space for water droplets to fit into it. At the dew point, the air cannot hold more water in its gaseous state. We know that water vapor is the gaseous form of water that contains water droplets. 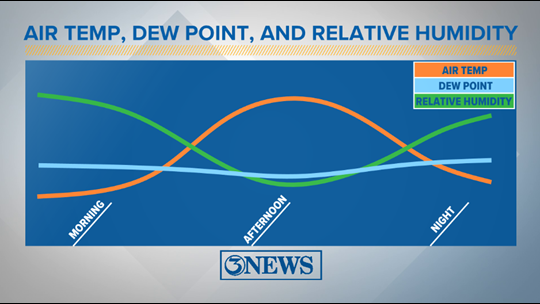 
Let's consider an example of water vapors. In the air, the condensation water is often known as cloud and fog. The condensation water, when it freezes, is known as frost, while on solid surface is known as dew. It means that below the dew point, the vapor condenses, and the rate of condensation becomes greater than that of the evaporation. The temperature below the dew point temperature causes the vapors to reach the condensation state. It is defined as the temperature point at which the saturation vapor pressure (maximum pressure by the vapors at a given temperature) equals the present vapor pressure. The dew point is related to the vapor pressure of the liquids.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
